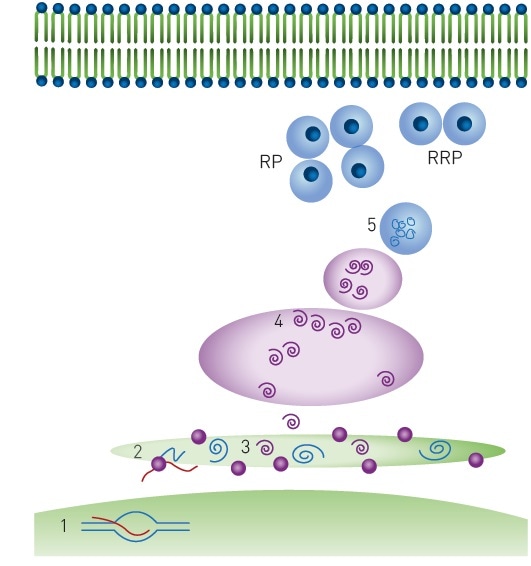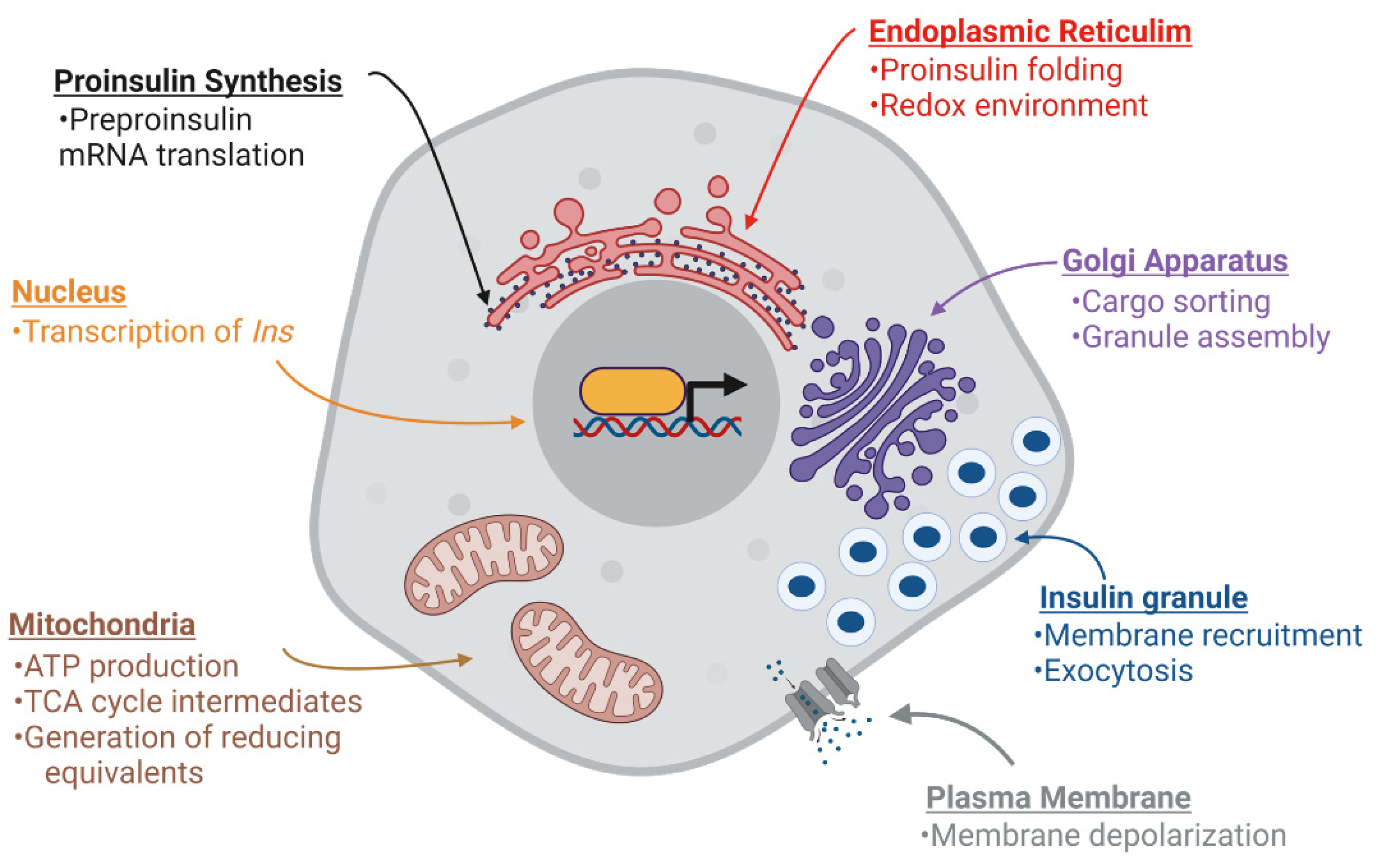
Biomolecules | Free Full-Text | Nutrient Regulation of Pancreatic Islet β-Cell Secretory Capacity and Insulin Production

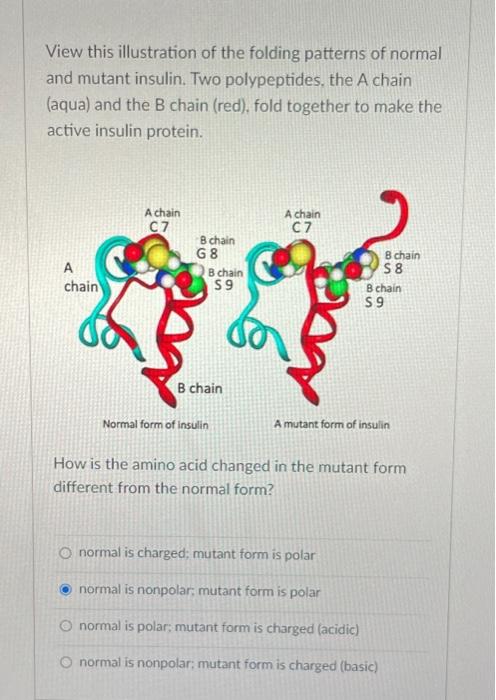
The Folding Nucleus of the Insulin Superfamily: A FLEXIBLE PEPTIDE MODEL FORESHADOWS THE NATIVE STATE - ScienceDirect
![PDF] Sequences of B-chain/domain 1-10/1-9 of insulin and insulin-like growth factor 1 determine their different folding behavior. | Semantic Scholar PDF] Sequences of B-chain/domain 1-10/1-9 of insulin and insulin-like growth factor 1 determine their different folding behavior. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/52f5c8fb5482353a9ecc0f313129195018c6ca26/2-Figure1-1.png)
PDF] Sequences of B-chain/domain 1-10/1-9 of insulin and insulin-like growth factor 1 determine their different folding behavior. | Semantic Scholar

Biomimetic Synthesis of Lispro Insulin via a Chemically Synthesized “Mini-Proinsulin” Prepared by Oxime-Forming Ligation | Journal of the American Chemical Society

Molecular Aspects of Insulin Aggregation and Various Therapeutic Interventions | ACS Bio & Med Chem Au
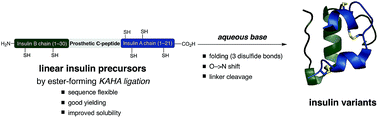
Facile folding of insulin variants bearing a prosthetic C-peptide prepared by α-ketoacid-hydroxylamine (KAHA) ligation - Chemical Science (RSC Publishing)

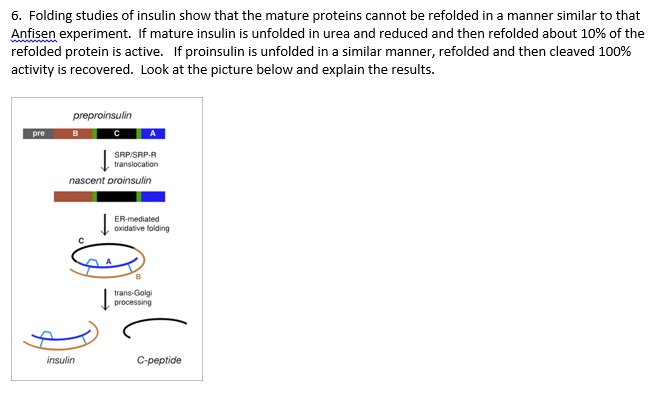
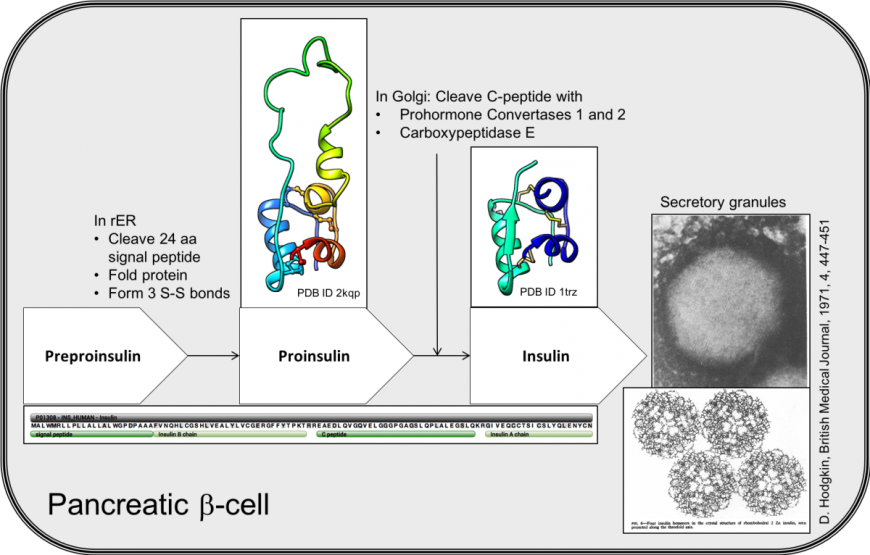
Biosynthesis, structure, and folding of the insulin precursor protein - Liu - 2018 - Diabetes, Obesity and Metabolism - Wiley Online Library

Insulin Biosynthesis, Secretion, Structure, and Structure-Activity Relationships - Endotext - NCBI Bookshelf
![Design and Folding of [GluA4(OβThrB30)]Insulin (“Ester Insulin”): A Minimal Proinsulin Surrogate that Can Be Chemically Converted into Human Insulin - Sohma - 2010 - Angewandte Chemie International Edition - Wiley Online Library Design and Folding of [GluA4(OβThrB30)]Insulin (“Ester Insulin”): A Minimal Proinsulin Surrogate that Can Be Chemically Converted into Human Insulin - Sohma - 2010 - Angewandte Chemie International Edition - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/8a9714ee-35cb-4fa5-a937-3eb7a4aae698/mfig001.jpg)
Design and Folding of [GluA4(OβThrB30)]Insulin (“Ester Insulin”): A Minimal Proinsulin Surrogate that Can Be Chemically Converted into Human Insulin - Sohma - 2010 - Angewandte Chemie International Edition - Wiley Online Library
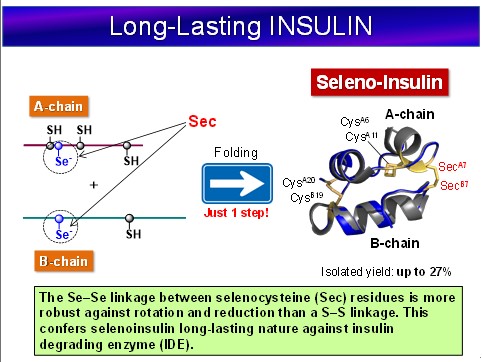
Experiments of Protein Folding | Tokai Univesity, Department of Chemistry, Iwaoka Laboratory | Iwaoka Laboratory

Mechanism of Insulin Fibrillation: THE STRUCTURE OF INSULIN UNDER AMYLOIDOGENIC CONDITIONS RESEMBLES A PROTEIN-FOLDING INTERMEDIATE - ScienceDirect